Improved SpO2 Accuracy with RD SET®
We Just Raised the Standard, Again
Home / Improved SpO2 Accuracy with RD SET™
Improved SpO2 Accuracy with RD SET™
Improved SpO2 Accuracy with RD SET®
Improved SpO2 Accuracy with RD SET®
We Just Raised the Standard, Again
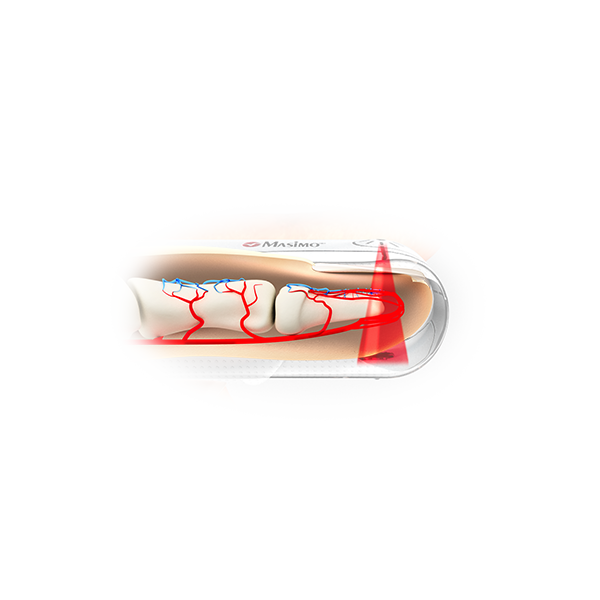
Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry is used to monitor more than 200 million patients a year1 and is the primary pulse oximetry technology at 9 of the top 10 hospitals according to the 2019-20 U.S. News and World Report Best Hospitals Honor Roll.2 Over 100 independent and objective studies have shown that Masimo SET® outperforms other pulse oximetry technologies.3 At Masimo, we never put innovation on hold – and our proven SET® technology is no exception.
Masimo is proud to announce that SpO2 accuracy has now improved in conditions of motion and no motion, providing clinicians with even greater confidence that the SpO2 values they rely on accurately reflect a patient’s physiological status.
SpO2 accuracy specifications have improved to 1.5% ARMS for all patients in conditions of motion and no motion.
SpO2 ARMS Accuracy for RD SET® Sensors (70-100%)
| Previous | Improved | |
| Motion | Previous Masimo SET®: 3% | Improved Masimo SET®: 1.5% |
| No Motion | Previous Masimo SET®: 2% | Improved Masimo SET®: 1.5% |
Improved SpO2 accuracy specifications are now available on the following RD SET sensors:
| Product Description | Weight Range |
| RD SET Adt, Adult SpO2 Adhesive Sensor | Weight Range:> 30 kg |
| RD SET Pdt, Pediatric SpO2 Adhesive Sensor | Weight Range:10-50 kg |
| RD SET Inf, Infant SpO2 Adhesive Sensor | Weight Range:3-20 kg |
| RD SET Neo, Adult/Neonatal SpO2 Adhesive Sensor | Weight Range:< 3 kg or > 40 kg |
| RD SET NeoPt, Neonatal SpO2 Adhesive Sensor | Weight Range:< 1 kg |
| RD SET NeoPt-500, Neonatal SpO2 Adhesive Sensor | Weight Range:< 1 kg |

Raise the Standard For Your Patients
Raise the Standard For Your Patients
Masimo SET® Solutions
References:
- 1.
Estimate: Masimo data on file.
- 2.
http://health.usnews.com/health-care/best-hospitals/articles/best-hospitals-honor-roll-and-overview.
- 3.
Comparative studies include abstracts presented at scientific meetings and peer-reviewed journals. Clinical studies on pulse oximetry and the benefits of Masimo SET® can be found at http://www.masimo.com/clinical-evidence.
RESOURCES
RD SET Sensors with improved SpO2 accuracy specifications are not licensed for sale in Canada.
For professional use. See instructions for use for full prescribing information, including indications, contraindications, warnings, and precautions.
PLCO-004075/PLM-11742B-0620